In 2006, I became interested in gasification as a way of generating energy from biomass while storing atmospheric carbon in the ground. I thought I would explain some of the experiments I did and some of the interesting things I found out.
What is gasification?
Gasification is the process of turning biomass, such as wood, into a fuel gas that an internal combustion engine can run on. Complete combustion of biomass produces water and carbon dioxide, but by restricting the amount of air allowed into the reactor you can produce an incompletely combusted gas made up of carbon monoxide, methane and hydrogen. This can then be piped into a normal spark ignition engine and used similarly to LPG.
There are four steps in gasification:
- Drying - The fuel is heated and water is removed from the biomass;
- Pyrolysis - The fuel heated without any oxygen breaks down and forms small volatile compounds (called tar or bio-oil) and solid charcoal;
- Combustion - The tar and charcoal are burnt in a small amount of oxygen from the air, generating heat for the entire process;
- Reduction - The amount of oxygen quickly runs out and the water and carbon dioxide are reacted on the hot charcoal surface to produce carbon monoxide and hydrogen.
 |
| Photo Credit: GEK |
Some of the benefits of gasification, as opposed to combustion on an open fire, includes the increased fuel efficiency, as combustion is much more efficient and clean when using a gas instead of a solid fuel. The conversion of biomass to electricity using simple combustion requires steam turbines which are only economical on a large scale. The ability to power an engine that can drive a generator means it is also a low-cost method to generate small scale power. By adding the charcoal that is generated to the soil, the entire process can be carbon negative by trapping the CO2 the tree took in during its growth and locking it away in a stable form of carbon charcoal.
These types of gasifiers were heavily deployed (in over a million vehicles) in Europe during WWII when fossil fuels were in limited supply. My favourite photo from this time is a picture of a tank powered by a gasifier.
 |
| Photo credit |
I built two different types of gasifiers - a gasifier stove and a downdraft gasifier, both of which I will outline below.
Gasifier stove
The first gasifier I built was a gasifier stove. The geometry of the gasifier is called a top-lit updraft gasifier (TLUD). This means the fuel is combusted from the top with the air moving up through the fuel. The diagram below shows the working principles.
 |
| Photo credit |
The fuel is lit from the top and air is supplied from the bottom. A flame front (migrating pyrolytic front) moves down through the fuel. The tar and water are pulled through the hot bed of coals, helping to break down some of the tar. Secondary air is then injected into the top of the reaction chamber which allows the fuel gas to burn cleanly. The stove was built from a computer supply box and used a forced draft from a computer fan which I powered on 12V DC. I mainly ran the stove on wood chips but I also used it to test the heat content of different fuels by heating water placed on top of the stove.
The stoves are not just a curiosity; thousands of them are being built and used in developing countries to improve the air quality for those who rely on solid fuels for cooking. The video below explains.
I also made use of the stove to study the combustion of algae during a summer research project working with Dr Rupert Craggs from National Institute of Water and Atmospheric Research (NIWA) in New Zealand. The algae were grown in open raceway ponds which used waste water to feed the algae.
 |
| Photo Credit: NIWA |
I made use of a non-woven geotextile to dry the algae from the 98% water content down to 7-12 wt% which is suitable for combustion. The higher heating value for the algae was 23.06 MJ/kg compared with wood at 14-17 MJ/kg. The dried algae formed flakes which made for excellent fuel and allowed for combustion in the gasifier stove. One thing I didn't measure was the emissions, as the high nitrogen content would suggest a large amount of nitrous oxide could be generated.
We published the results in a conference proceedings in 2010. https://www.waternz.org.nz/Article?Action=View&Article_id=786. In particular, we looked at the potential for algae to be carbonised to biochar to be a stable carbon sink.
Discovery model gasifier
The discovery model gasifier is a downdraft gasifier. This means the air is injected in the bottom and drawn down. The design of the gasifier is based on the Pacific class gasifier from a New Zealand company called Fluidyne. I scaled it down so that it could power a 660 cc engine at 1500 rpm outputting 3 kW of energy. This required a gas output of 9024 m3/hr of wood gas with a wood consumption of 4.19kg/hr. I initially had a 1kg hopper which allowed for a short test run of around 20 minutes. The design of the gasifier is really quite interesting and was designed to be built at a very low cost (a diagram of the gasifier is shown below). The fuel is loaded into the top and moves down as it is consumed. The fuel is dried and is broken down to tar and charcoal in the pyrolysis zone. Air is then injected through three nozzles and allows for combustion. A tube then comes up through the charcoal into the oxidation zone. Many gasifier designs make use of a metal throat that mechanically constricts the fuel. However, this throat can melt as the high temperatures are hot enough to melt steel. This gasifier makes use of the charcoal itself to act as the throat and the insulation allowing for low-cost materials to be used. As the carbon dioxide and water enter the tube the hot charcoal, in the absence of air, produces carbon monoxide and hydrogen.
The biggest advantage of a downdraft gasifier design is that all of the tar must go through the oxidation zone and then the reduction zone. This makes the fuel gas generated from these types of gasifiers very clean.
Here is picture inside the reactor with the constriction tube and the nozzles (the bolts are being stored there and are not used during operation). You can also see the diesel glow plug I used to start the gasifier in the top right corner.
The fuel I used was mainly wood chips or small wood rounds from the garden. The fuel gas then passed through a series of cleaning stages to prepare it for the engine. I used a blast tube to remove the large particles and some of the soot. A cyclone particle separator was made to remove the micron-sized soot particles. Cooling tubes were used to condense the water out of the gas and to generally reduce the temperature of the gas as well as to increase the density of the fuel gas. Finally, it went through a sawdust filter to remove any particles or tar that were missed in the previous stages. I later replaced the sawdust filter with a bag filter which could be cleaned and reused.
The gasifier was designed for a large generator, which I didn't end up finishing, but I did some preliminary tests with a smaller generator. Here is an interview I did where I started up the gasifier and ran the engine.
One of the important aspects of making the gasifier work well (i.e. tar free) was to adjust the height of the reduction tube so that it was inside the oxidation zone and the grate height to allow the fuel to flow. Two good checks for a tar free operation was a blue flame (meaning no hydrocarbons in the fuel) and no hydrocarbons in the condensate from the fuel gas cooler. As you can see from the picture above I got close to correctly tuning the gasifier however Fluidyne's Andes class gasifier flaring shows a really excellently tuned gasifier with only carbon monoxide and hydrogen burning.
 |
| Fluidyne |
The microlab gasifier was built by Fluidyne in 2011 and is the same size as the discovery model gasifier but with two cyclones and is now being used for research at the University of Ulster.
 |
| Microlab gasifier |
I have Doug Williams from Fluidyne to thank for showing me how to build and operate gasifiers. I also have to thank Peter Wilkinson from Wilkinson Transport Engineers, who allowed me to use his workshop and materials to build the gasifier.
Continued interest
My PhD research is on combustion, global warming and reducing soot emissions from engines so this still interests me greatly. Gasification of biomass is one of the key technologies for controlling the amount of carbon dioxide in the atmosphere. This is often referred to as bioenergy, with carbon capture and storage (BECCS). CO2 is captured by trees and the CO2 released during burning can be stored, making the process carbon negative.
 |
| Photo Credit: Drax Power |
A second option is to burn some of the carbon to CO2 and to store the rest of the carbon as solid charcoal. This is called bioenergy-biochar systems (BEBCS). This does not sequester all of the carbon but as the charcoal is easier to handle and when added to the soil (referred to as biochar) can improve the holding of nutrients. This process is cheaper as the biochar can be sold to offset the cost.
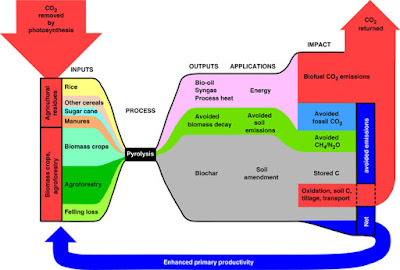 |
| Photo credit |
A recent paper on BEBCS compared with BECCS showed that in the short term, the former would be easier and cheaper to deploy. Biochar added to the soil also significantly reduces the amount of nitrous oxide emitted (as nitrous oxide comes from fertilisers and is ~300 times more potent a greenhouse gas than CO2).
I will probably be writing more about biochar in the future, but feel free to ask any questions about gasifiers.







































